11th July 2020, Dr Chee L Khoo

Another month, another change to the immunisation schedule. You must have heard about the major changes to schedule from 1st July. There are changes to who should get the pneumonia vaccines and which pneumonia vaccine they should get under the National Immunisation Program (NIP). Oh, that is, if they don’t run out of stocks again. There are also major changes to who gets which meningococcal vaccines and when. I don’t know about you but I am totally confused. Print off revised easy to follow fridge magnets below. If you are not confused about the new changes, then read no further.
Pneumococcal vaccine schedule
Is it not strange that we are going to a 13 valent vaccine first before the 23 valent vaccine? There are two types of pneumococcal vaccines – pneumococcal conjugate vaccine (PCV) and pneumococcal polysaccharide vaccine (PPV). Pneumovax 23 is a 23-strain pneumococcal conjugate vaccine (23vPPV) while Prevenar 13 is a 13-strain pneumococcal polysaccharide vaccine (13vPCV). Estimates of efficacy and effectiveness for 23vPPV are lower and more variable than for 13vPCV, particularly against community acquired pneumonia (CAP) outcomes.
23vPPV induces significant immune responses in immunocompetent adults. Immune responses are similar in older people (aged 70–80 years) and younger people (aged 50–60 years), but poorer in people who are immunocompromised.
The vaccine effectiveness of 3 doses of 13vPCV against invasive pneumococcal disease (IPD) due to 13vPCV serotypes was estimated at 86% for non-Indigenous children. The Community Acquired Pneumonia Immunisation Trial in Adults (CAPiTA) study showed that 13vPCV was efficacious against pneumococcal disease in vaccine-naive adults aged ≥65 years. Efficacy was 46% against a 1st episode of vaccine-type CAP and 75% against a 1st episode of vaccine-type IPD.
Sequential administration of 13vPCV and 23vPPV in people with risk conditions is likely to protect against 23v-non-13v serotypes.
Children
Healthy kids < 12 months old (non-indigenous as well as ATSI children):
Unchanged i.e. Prevenar 13 (13vPCV) at 2, 4 and 12 months (3 doses in total)
Click here for Healthy Kids Schedule Fridge Magnet
Kids with medical risks
<12 months old (i.e. babies):
- Prevenar 13 (13vPCV) at 2, 4, 6 and 12 months (4 doses in total) followed by
- Pneumovax 23 (23vPPV) at 4 years and
- Pneumovax 23 (23vPPV) at 9 years old (2 doses in total)
Click here for Kids with Medical Risks Schedule Fridge Magnet
>12 months old (i.e. chronic disease diagnosed later)
- Prevenar 13 (13vPCV)) at diagnosis followed by
- Pneumovax 23 (23vPPV) 2-12 months later or at 4 years old followed by
- Pneumovax 23 (23vPPV) 5 years later
ADULTS
Healthy Non-indigenous adults >70 years old
Prevenar 13 (13vPCV)
(Used to be Pneumovax 23 at 65 years old and then every five years)
All ATSI adults > 50 years old
- Prevenar 13 (13vPCV) at diagnosis followed by
- Pneumovax 23 (23vPPV) 2-12 months later followed by
- Pneumovax 23 (23vPPV) 5 years later
Adults with medical risks
- Prevenar 13 (13vPCV)) at diagnosis of chronic illness followed by
- Pneumovax 23 (23vPPV) 2-12 months later or at 4 years old followed by
- Pneumovax 23 (23vPPV) 5 years later
Click here for Adults with or without Medical Risks Schedule Fridge Magnet
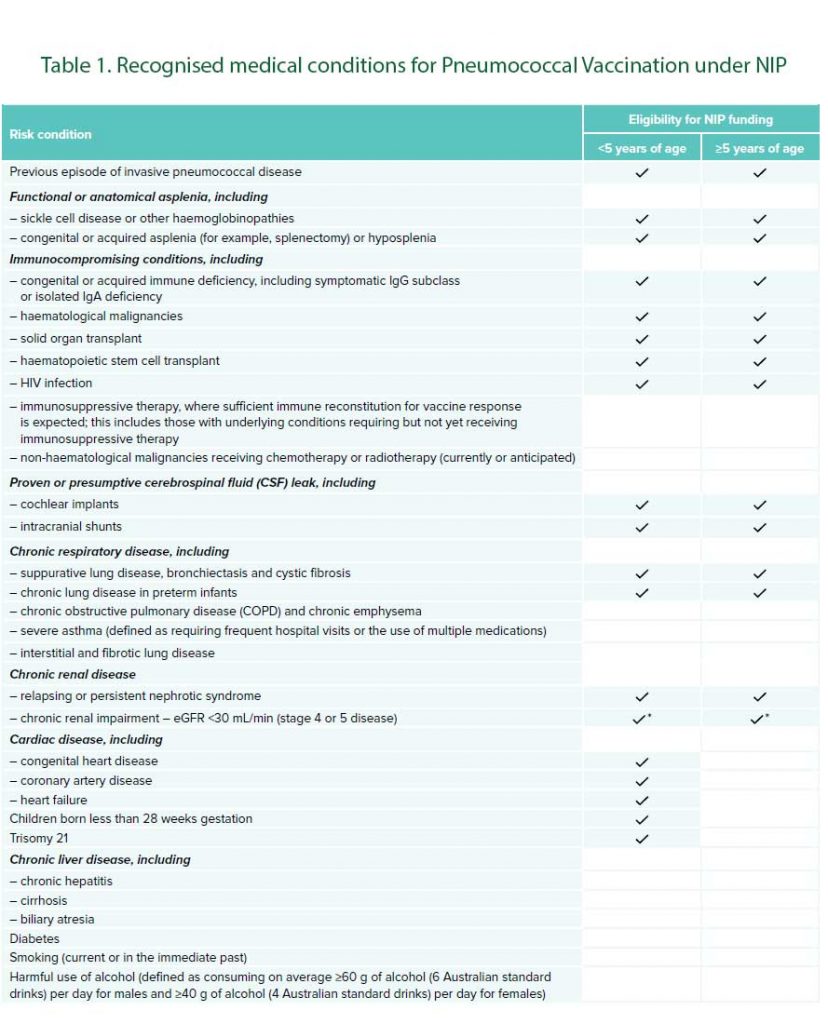
See Table 1 for list of recognised medical conditions for pneumococcal vaccination
Summary of changes for pneumococcal vaccination
- Normal kids – unchanged i.e. 3 doses of Prevenar 13
- Kids with medical risks – 4 doses of Prevenar 13 PLUS 2 doses of Pneumovax 23
- Healthy Adults >70 yo – one dose of Prevenar 13
- Adults with medical risks – Prevenar 13 followed by Pneumovax 2-12 months later then Pneumovax 5 years later
- ATSI Adults >50 yo – Prevenar 13 followed by Pneumovax 2-12 months later then Pneumovax 5 years later
Meningococcal vaccination schedule
We have Bexsero (meningococcal B) and Nimenrix (meningococcal ACWY) vaccines.
Non-indigenous children with no specific medical conditions (see below)
Unchanged
- Nimenrix (meningococcal ACWY) continues to be given at 12 months old followed by
- Nimenrix (meningococcal ACWY) at 14-16 years old – through school program
Aboriginal and Torres Strait Islander children with no specific medical conditions (see below)
- Bexsero (meningococcal B) 1 dose at 2, 4 and 12 months of age (3 doses in total).
- Nimenrix (meningococcal ACWY) continues to be given at 12 months old
- Nimenrix (meningococcal ACWY) at 14-16 years old – through school program
Click here for Healthy ATSI kids schedule Fridge Magnet
Aboriginal and Torres Strait Islander children at risk for invasive meningococcal disease (IMD)
- Bexsero (meningococcal B) 1 dose at 2, 4, 6 and 12 months of age (4 doses in total).
- Nimenrix (meningococcal ACWY) continues to be given at 12 months old
- Nimenrix (meningococcal ACWY) at 14-16 years old – through school program
Click here for ATSI Kids at risk of IMD Schedule Fridge Magnet
Bexsero Catch up for Aboriginal and Torres Strait Islander children
Bexsero (meningococcal B) vaccine catch-up is available for all Aboriginal and Torres Strait Islander children aged <2 years (i.e. up to 23 months) for the first three years of the program (i.e. until 30 June 2023). The dose schedule will depend on the age when the catch-up vaccination is initiated.
Adults:
Both the Bexsero (meningococcal B) and Nimenrix (meningococcal ACWY) are funded under the NIP for people of all ages with some medical conditions that increase their risk of IMD.
See Table 2 for the specific medical conditions for Bexsero vaccination under the NIP.

Implications of the new schedule:
New schedule for pneumococcal vaccination for the newborn
New schedule for pneumococcal for those with medical risks
New schedule for patients with ATSI background
Catch up for those with newly diagnosed medical risks
Catch up for those >70 who had pneumovax 23 previously
