15th September 2018, Dr Chee L Khoo
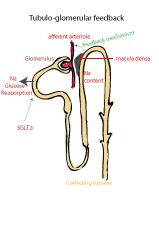
Glomerular Hyperfiltration
Although the physiological factors that cause early diabetic renal injury remain incompletely understood, glomerular hyperfiltration (GH) has been associated with the development of diabetic nephropathy (DN). GH leads to renin-angiotensin-aldosterone system (RAAS) activation which in turn leads to systemic vascular and endothelial abnormalities. DN not only lead to dialysis and ultimately, renal transplantation in some, it is a leading cause of mortality in patients with diabetes. Sadly, in patients with T1D, a quarter of those with DN will progress to end stage renal failure.
Apart from early diagnosis, tight glycaemic and BP control, the inhibition of the RAAS continues to be the mainstay therapy for the prevention and treatment of DN. However, the reno-protective role of ACEi/ARBs has recently been challenged by data from the Renin Angiotensin System Study (RASS). Early primary prevention with ACE inhibition or angiotensin receptor blockade did not modify DN progression in adults with T1D (17). Similar large clinical trials including ONTARGET, ALTITUDE and VA-NEPHRON-D, have also failed to demonstrate improved renal outcomes in diabetic subjects with dual RAAS inhibition using combined ACEi and ARB or using direct renin inhibitor-related regimens (18).
Oxidative stress, Nox and DN
Hyperglycaemia leads to oxidative stress which is now recognised as a critical mechanism in the pathogenesis of microvascular complications including DN. Oxidative stress leads to increased reactive oxygen species (ROS) generation. Numerous studies have identified the Nox family of NAPDH oxidases as a major source of ROS. All 5 of the Nox family members have been found in renal tissues but Nox1 and Nox4 are most studied. Nox1 and Nox4 are closely related.
Evidence from animal model studies suggest that many of the agents that are harmful to the glomeruli work primarily through the Nox 4 derived ROS system. These toxic agents either increase Nox 4 protein expression, increase activators of Nox 4 or upregulation of the Nox 4 mRNA. For example, Angiotensin II elicits increases in Nox 4 protein expression and upregulation of Nox 4 mRNA in the mesangial cells. Similarly, induction of glomerular injury by AGEs appears to be mediated by Nox related ROS.
See “Meet your noxious cousins from the Nox family”
The discovery of the central role of these Nox homologues, in particular Nox 4 and Nox 1, have triggered the development of Nox inhibitors. Pre-clinical studies performed with these inhibitors in experimental animal models indicate that they effectively attenuate the pathological changes observed in renal complication of type 1 and type 2 diabetes, atherosclerosis, ischemic retinopathy, liver fibrosis and idiopathic pulmonary fibrosis. In other words, if we can somehow block these Nox homologues, can we prevent further renal injury in patients with T1D?
Mice treated with a Nox 1/Nox 4 inhibitor, GKT137831 had reduced glomerular hypertrophy, mesangial matrix expansion, urinary albumin excretion, and podocyte loss. Another inhibitor, GKT136901 was found to ameliorate DN in db/db mice, a model of type 2 diabetes.
One of those inhibitors, GKT137831 have recently been successfully used in a phase 1 clinical trial. In a short-term phase 2 trial in patients with type 2 diabetes and kidney disease, GKT37831 demonstrated an excellent safety profile and achieved statistically significant reductions in several secondary efficacy endpoints. However, improvements in albuminuria, the study’s primary efficacy endpoint, was not achieved after 12 weeks of treatment.
A further phase 2 clinical trial in patients with type 2 diabetes and albuminuria is now being evaluated. The Baker Institute study is a placebo-controlled, double blind, randomised, parallel group phase 2 trial designed to evaluate the effect of oral GKT831 on the urine albumin-to-creatinine ratio (UACR) in patients with type 1 diabetes and persistent albuminuria despite treatment with optimal standard of care.
The primary endpoint of the study will be UACR difference at the end of treatment period of 48 weeks. A key secondary endpoint of the study will be the effect of GKT37831 on changes in estimated glomerular filtration rate. They are recruiting 142 patients from up to 15 investigational centres in Australia. Campbelltown is one of those centres. You don’t often see a major drug trial coming to a centre near you!
While almost all new young T1D are now comprehensively managed in multi-disciplinary centres, we still have many older T1D that are managed in primary care in conjunction with our endocrinology colleagues. A significant proportion of these will have diabetic nephropathy. They may be very suitable for this very important and exciting trial.
If you have a suitable patient, contact:
Maureen Mattick
Research Co-ordinator: Macarthur Clinical School
P: 46344594| F:46203166| E: baker@westernsydney.edu.au
Reference
Mauer M, Zinman B, Gardiner R, Suissa S, Sinaiko A, Strand T, et al. Renal and retinal effects of enalapril and losartan in type 1 diabetes. The New England journal of medicine. 2009; 361(1):40–51.
Mann JF, Anderson C, Gao P, Gerstein HC, Boehm M, Ryden L, et al. Dual inhibition of the renin-angiotensin system in high-risk diabetes and risk for stroke and other outcomes: results of the ONTARGET trial. J Hypertens. 2013; 31(2):414–21.
Gorin Y et al. Targeting NADPH oxidase with a novel dual Nox1/Nox4 inhibitor attenuates renal pathology in type 1 diabetes. Am J Physiol Renal Physiol. 2015 Jun 1;308(11):F1276-87
Andrzej S. Krolewski, MD, Tomohito Gohda, Monika A. Niewczas. Progressive Renal Decline as the Major Feature of Diabetic Nephropathy in T1D. Clin Exp Nephrol. 2014 August ; 18(4): 571–583.
Petter Bjornstad, David Cherney, David M. Maahs. Early Diabetic Nephropathy in Type 1 Diabetes – New Insights. Curr Opin Endocrinol Diabetes Obes. 2014 August ; 21(4): 279–286
https://www.businesswire.com/news/home/20170628006005/en/Genkyotex-Announces-Investigator-Initiated-Phase-2-Clinical-Trial
