26th May 2019, Dr Chee L Khoo
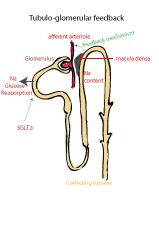
Sometimes when a drug is contraindicated for patients with low eGFR, it may mean that usage of the drug may further damage the kidneys or increase the risk of certain adverse events. For example, the use of metformin in patients with eGFR <30 significantly increase the risk of lactic acidosis. Usage of lithium in patients with impaired renal function (low eGFR) may further damage the kidneys. Sometimes, the drug is “contraindicated” because under a certain eGFR, the efficacy of the drug is reduced. This is the case with SGLT2 inhibitors (SGLT2i). But does the use of SGLT2i in these patients damage the kidneys if used in low eGFR conditions?
SGLT2i were designed to improve blood glucose by reducing the re-absorption of glucose from the renal tubules in patients with diabetes. To meet regulatory requirements, a number of landmark cardiovascular outcome trials have demonstrated safety in patients with high cardiovascular risks and benefit in patients with established cardiovascular events (DECLARE-TIMI 58, EMPA-REG, CANVAS). Subsequent analyses of these trials suggested that SGLT2 inhibition might improve renal outcomes; however, the number of patients with significant renal impairment were low and only a relatively few patients reached end-stage kidney disease.7-9 Remember, these were cardiovascular outcome trials but happen to show potential renal benefits. We needed trials that are specifically designed to assess the effects of SGLT2i on renal outcomes in patients with diabetes and significant renal impairment e.g. albuminuric chronic kidney disease.
The first of these trials, CREDENCE published their results in April 2019 (1). 4401 patients with type 2 diabetes, HbA1c between 6.5-12%, eGFR 30-90 ml/min/1.73m2 and albuminuria were randomised to receive either canagliflozin 100mg or placebo. All patients received a stable dose of ACE/ARB for at least 4 weeks. The primary outcome was end stage renal disease (dialysis, renal transplant or eGFR <15ml/min/1.73m2, doubling of creatinine levels, death from renal or cardiovascular disease).
There were many secondary outcomes: 1) composite of cardiovascular death or hospitalisation for heart failure; 2) composite of cardiovascular death, myocardial infarction or stroke; 3) hospitalisation for heart failure, 4) composite of end-stage kidney disease, doubling of the serum creatinine level, or renal death; 5) cardiovascular death; 6) death from any cause; and 7) a composite of cardiovascular death, myocardial infarction, stroke, or hospitalisation for heart failure or for unstable angina.
After a median follow up of 2.6 years, there was a 30% reduction in the primary event rate in the canagliflozin group compared with placebo. The relative risk of the composite of end-stage kidney disease,doubling of the serum creatinine level, or renal death was lower by 34% in the canagliflozin group. Canagliflozin was also associated with lower risk of the composites of cardiovascular death or hospitalization for heart failure (hazard ratio, 0.69; 95% CI, 0.57 to 0.83; P<0.001), cardiovascular death,myocardial infarction, or stroke (hazard ratio,0.80; 95% CI, 0.67 to 0.95; P = 0.01), and hospitalization for heart failure.
These results indicate that canagliflozin, an SGLT2i may be an effective treatment option for renal and cardiovascular protection in patients with type 2 diabetes with chronic kidney disease. The improvement in cardiovascular outcomes is consistent with the improvement seen with Dapagliflozin (Forxiga) and Empagliflozin (Jardiance) in the DECLARE-TIMI 58 and EMPA-REG OUTCOME trials (2-4).
The CREDENCE trial is the first of many renal outcome trials with SGLT2i. DAPA-CKD (dapagliflozin) is due to report in November 2020. EMPA-Kidney (empagliflozin) is due to report in June 2022.
Access the abstract here.
References:
- Vlado Perkovic, Meg J. Jardine, Bruce Neal, et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. NEJM April 14, 2017.
- Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017; 377: 644-57.
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373: 2117-28.
- Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2019; 380: 347-57.
