29th March 2026, A/Prof Chee L Khoo

Just as we are getting used to the GLP1-RA and the bi-hormonal GLP1/GIP co-agonists in the management of type 2 diabetes (T2D) and obesity, the playground is about to get more crowded. Retatrutide is a once weekly triple hormone receptor agonist. It is a single molecule that can activate the receptors for glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon. The Phase 3 trial result was just released. It demonstrated incredible efficacy in glucose lowering and weight loss. Before long, it will be in Australia alongside the other incretin mimetics.
Using a glucagon agonist to treat T2D? Doesn’t glucagon increase plasma glucose levels? Let’s look at how a glucagon receptor (GCGR) agonist can join the team for glucose lowering and weight loss. We know that glucagon opposes the action of insulin and increases blood glucose levels by promoting glycogenolysis.
Thus, theoretically, activation of the glucagon receptor will lead to an increase in plasma glucose levels. However, in practice, the situation is a lot more complicated than that. All the glucagon agonists come paired with either a GLP1 agonist or a GIP agonist or both. In preclinical animal models, glucagon agonism has been shown to reduce food intake and to increase energy expenditure, contributing to weight loss (1). The potential in increasing plasma glucose is negated by the powerful GLP1 and/or GIP agonist that the glucagon agonist comes with.
Bi-hormonal GLP-1R/GCGR co-agonists works by utilising glucagon’s powerful promotion of energy expenditure and GLP-1’s anorectic and glucose-lowering effects. As mentioned, the presence of GLP-1R agonism minimise potential glycaemic effects of GCGR agonism.
Retatrutide is a single molecule which has domains on the same molecule that that activate the GLP1, GIP and Glucagon receptors. The integration of glucagon-mediated energy expenditure with the appetite-suppressive and insulinotropic effects of GIP and GLP-1 agonism is hypothesised to explain the substantial weight loss and metabolic improvements observed in Phase 2 trials.
TRANSCEND-T2D 1 trial just reported its topline results 10 days ago (2). We don’t have the full results yet. They will be presented at the American Diabetes Association (ADA) meeting in June 2026.
TRANSCEND-T2D 1 enrolled adults diagnosed with type 2 diabetes with inadequate glycaemic control with diet and exercise alone and a mean duration of diabetes of 2.5 years. The baseline HbA1c was 7.9%. In the primary end point, after 40 weeks, there was a 1.7 – 2.0 % reduction in HbA1c compared with 0.8% in the placebo arm. The key secondary endpoint was percentage weight loss from a baseline 96.9kg (BMI 35.8 kg/m2). There was up to a -16.8% weight reduction compared with -2.6% in the placebo arm. See table 1.
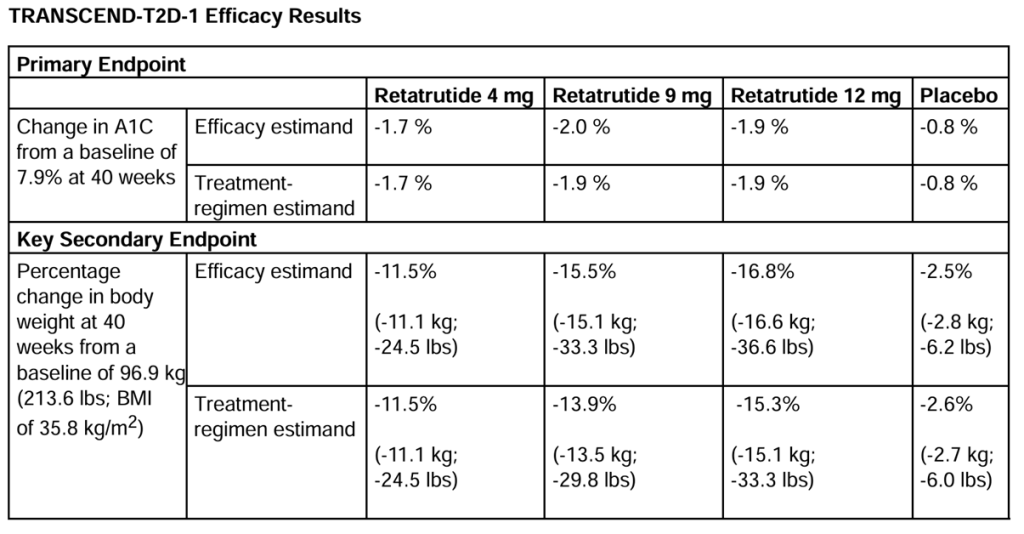
Efficacy estimand = analysis of only participants who remained in the trial to the end
Treatment estimand = analysis of all participants including those who dropped out
There were also improvements in the key cardiovascular risk factors including triglycerides, non-HDL cholesterol and systolic blood pressure.
The adverse effects were similar to other incretin therapies. Discontinuation rates due to adverse events were 2.2%, 4.5% and 5.1% with retatrutide 4 mg, 9 mg and 12 mg, respectively, compared to 0.0% with placebo.
This is the first of the many TRANSCEND trial series to come. This is the first time a tri-hormonal agonist have demonstrated in a phase 3 trials the efficacy in glucose lowering and weight reduction.
The weekly GLP1/glucagon agonist, survodutide has reported positive results in a phase 2 trial with up to 1.7% reduction in HbA1c and up to -8.7% in body weight after 16 weeks (3). In the meantime, the monthly MariTide (maridebart cafraglutide) has also reported positive results in its phase 2 trials with up to -20% weight reduction (4).
With the oral GLP1-agonists coming (probably) sometime this year, the market will be very crowded soon. By the way, generics of semaglutide will be also coming this year as the patent has expired in some countries (5).
References:
- Conceição-Furber E, Coskun T, Sloop KW, Samms RJ. Is glucagon receptor activation the thermogenic solution for treating obesity?? Front Endocrinol (Lausanne). 2022;13: 868037.
- https://investor.lilly.com/news-releases/news-release-details/lillys-triple-agonist-retatrutide-demonstrated-significant . Accessed 29/3/2026.
- Blüher M, Rosenstock J, Hoefler J, Manuel R, Hennige AM. Dose-response effects on HbA1c and bodyweight reduction of survodutide, a dual glucagon/GLP-1 receptor agonist, compared with placebo and open-label semaglutide in people with type 2 diabetes: a randomised clinical trial. Diabetologia. 2024 Mar;67(3):470-482
- https://www.amgen.com/newsroom/press-releases/2025/06/results-from-amgens-phase-2-obesity-study-of-monthly-maritide-presented-at-the-american-diabetes-association-85th-scientific-sessions. Accessed 29/3/2026.
- https://www.emarketer.com/content/novo-nordisk-missed-patent-deadline-canada–opening-door-generic-glp-1s-soon-2026
